iCura Diagnostics is conveniently located in Great Valley Industrial Park, the tri-state area outside of Philadelphia, and close to New Jersey, Delaware, as well as Maryland. The company provides diagnostic testing and servcies for the pharmaceutical industry, academic research center, and partner CRO and laboratories. iCura has a CLIA registered laboratory which has Pennsylvania Department of Health issued laboratory permit for clinical diagnostic tests as well. The company has a highly experienced team composed of board-certified pathologists and PhD level scientists, with active medical licenses and other professional certifications.
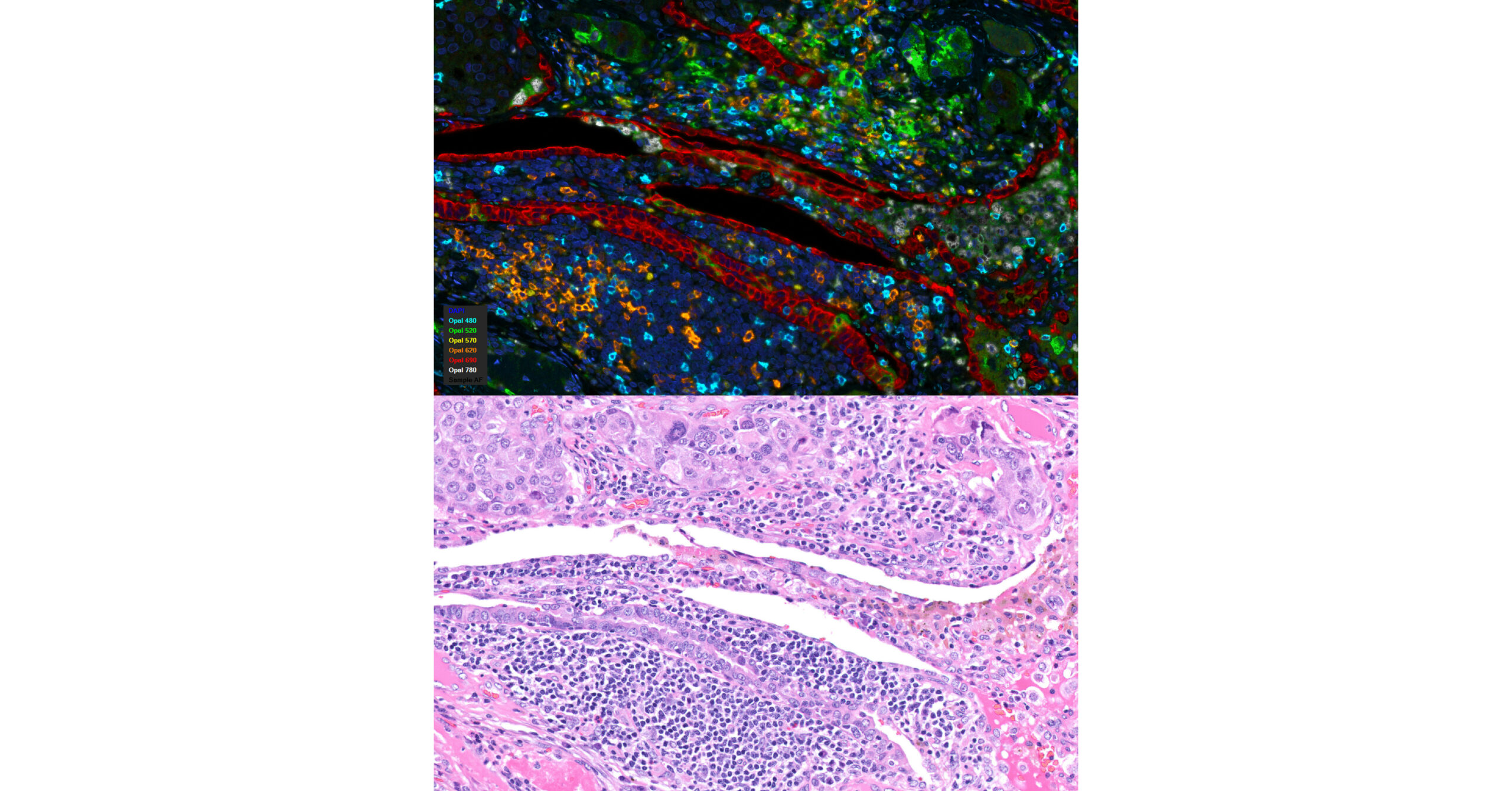
The details of the service items could be found on their website, www.icuradx.com, including digital pathology, whole slide imaging, immunohistochemistry, mutiplexing immunofluorescence, image analysis on cancer diagnosis, circulating tumor cells (CTC), biomarker discovery and development, and consultation services for laboratory certification, CLIA and CAP compliance, as well as laboratory permit applications. A full line of genomic services for oncology, microbiology, cardiovascular, as well as other genetic research, including, rtPCR, digital PCR, next generataion sequencing (NGS), is also under development. The company have obtained contracts from and performed services for major pharmaceutical companies, academic research centers, as well as other CRO and laboratory partners. For further information or potential interest for iCura’s service, users can fill out a client request form on the website, or call iCura directly at office: (484)5886055; mobile: (424)6452254.
SOURCE iCura Diagnostics , LLC