Hackensack Meridian Health, New Jersey’s largest and most comprehensive health network which has treated more COVID-19 patients than any other health system in the state, is now enrolling individuals in the Janssen Pharmacutical Companies of Johnson & Johnson’s Phase 3 COVID-19 vaccine trial.
The Phase 3 international, randomized, double-blind, placebo-controlled ENSEMBLE 2 trial run by Johnson and Johnson is designed to evaluate the safety and efficacy of a two-dose regimen of Janssen’s investigational COVID-19 vaccine candidate versus placebo in up to 30,000 adults 18 years old and older. The trial will include those both with and without comorbidities associated with an increased risk for severe COVID-19.
“We are proud to be at the forefront of history in creating safe and effective vaccines during this unprecedented global pandemic,’’ said Robert C. Garrett, FACHE, chief executive officer of Hackensack Meridian Health. “This is another example of Hackensack Meridian Health’s commitment to transforming health care in New Jersey and beyond.’’
“Vaccines are one of the major pathways to victory over COVID-19,” said Ihor Sawczuk, M.D., FACS, president of Hackensack Meridian Health’s Northern Market, and the chief research officer of the network. “Our health network is among those at the forefront of this fight.”
The Janssen COVID-19 vaccine candidate leverages the Company’s AdVac® technology platform, which was also used to develop and manufacture Janssen’s European Commission approved Ebola vaccine and construct its Zika, RSV, and HIV investigational vaccine candidates. Janssen’s AdVac® technology platform has been used to vaccinate more than 150,000 individuals to date in clinical studies across Janssen’s investigational vaccine programs.
This ENSEMBLE 2 trial involves a two-dose regimen, administered on day 1 and day 57 of participation. Half of the volunteers will be administered the vaccine, with the other half administered placebo. The total study duration is more than two years.
The two-dose ENSEMBLE 2 study will run in parallel to and separate from the ongoing Phase 3 ENSEMBLE trial, which is now fully enrolled with approximately 45,000 participants across three continents and evaluating a single-dose of the Janssen COVID-19 vaccine candidate. Conducting the ENSEMBLE and ENSEMBLE 2 trials in parallel will provide valuable insights into the dosing schedules and help determine a path forward in both the short term and the long term to potentially help end the fast-moving COVID-19 pandemic.
Hackensack Meridian Health investigators will enroll up to 300 of adults over 18 years of age from the New Jersey area.
“As the leading academic medical center in Monmouth and Ocean counties, Jersey Shore’s research team is well-suited to conduct this important medical research, which is crucial as we battle COVID-19,” said Kenneth N. Sable, M.D., MBA, FACEP, regional president, Hackensack Meridian Health, Southern Market.
Built on a legacy of purpose-driven actions and a commitment to diversity and inclusion, the Company aims to achieve representation of ethnically diverse populations and those that are over the age 60 in the implementation of its COVID-19 Phase 3 clinical trial program. For more information on why diversity in clinical trials is vital to the process, visit www.ResearchIncludesMe.com.
“We’re glad to play a part in this critical work,” said Ed Liu, M.D., chief of infectious diseases at Jersey Shore University Medical Center, and the study site’s principal investigator. “Since COVID-19 arrived, we have continually improved our ways to treat those affected by the virus, but having vaccine options could help us get past this pandemic.”
For more information about the trial, those interested can contact (551) 996-5977 or visit https://www.hackensackmeridianhealth.org/covid19/.
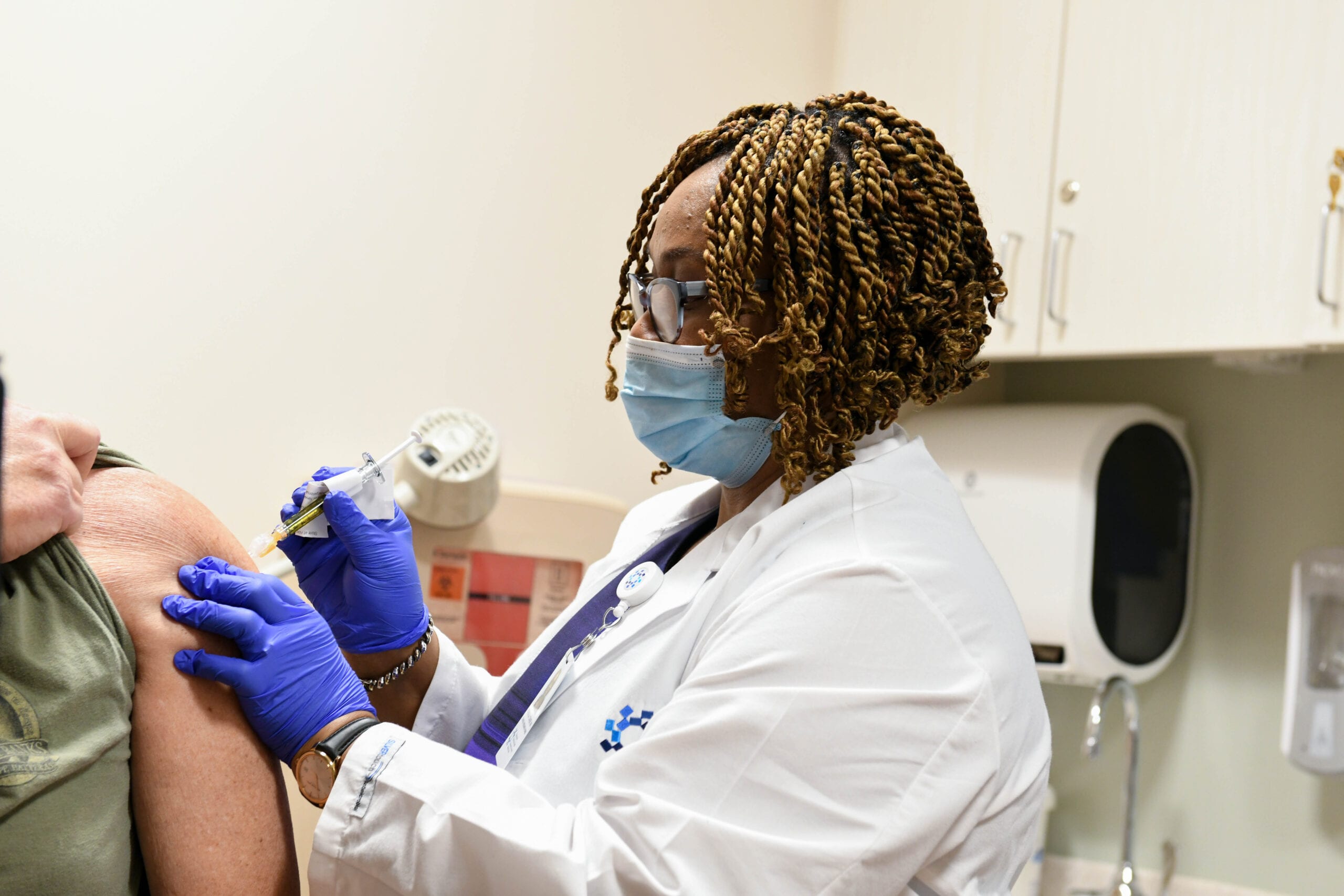
Photo caption:
Abimbola Coker, R.N., administers a shot at Jersey Shore University Medical Center as part of the first day of the Janssen/J&J ENSEMBLE 2 clinical trial for a two-dose COVID-19 vaccine.